Treatment options for central sleep apnoea
It can be challenging to treat patients with CSA, who often experience fewer or different symptoms than OSA patients. Adaptive servo-ventilation, as delivered by the AirCurve 10 CS PaceWave, is an appropriate choice for treating most of these more complex patients.*
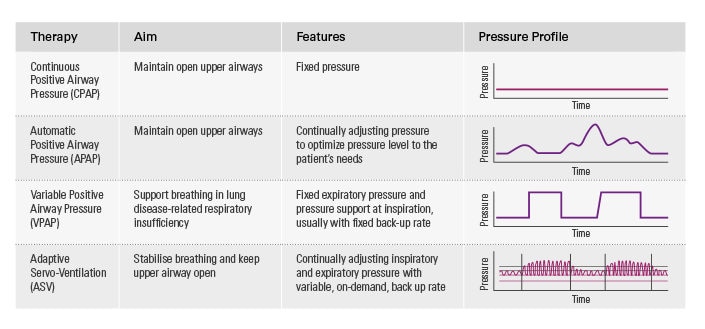
Central sleep apnoea treatment options
Continuous positive airway pressure or automatic positive airway pressure (CPAP/APAP) therapy is often used as the treatment of first intention, but experience shows that CPAP/APAP does not consistently control apnoeas or improve symptoms. As a result, some patients treated with CPAP/APAP remain symptomatic. This increases the risk of non-compliance and the likelihood that they will require multiple, resource-intensive interventions.
Adaptive servo-ventilation* is often an appropriate choice for treating these more complex patients, either as a first intention therapy or after an unsuccessful trial with other positive airway pressure (PAP) therapies.
Adaptive-servo ventilation: a proven solution for CSA
ASV is a form of PAP therapy that delivers auto-adjusting pressure support with automatic pressure support and expiratory positive airway pressure. ASV helps to treat both obstructive and central respiratory events and maintains adequate ventilation in response to patients’ changing needs.
Compared to other forms of PAP therapy, ASV offers significant benefits for the treatment of central sleep-disordered breathing.* This has been demonstrated in multiple clinical trials across various patient types.
More about treatment options

ASV clinical benefits
Compared to other forms of PAP therapy, ASV can offer significant benefits for the treatment of central SDB.* This has been demonstrated in multiple clinical trials across various patient types.

Treatment options for OSA
ResMed offers a range of effective treatment options for OSA, including CPAP, APAP, bilevel therapy and oral appliance therapy.

Digital health for sleep apnoea
ResMed connected solutions enable pro-active monitoring by healthcare professionals and result in more responsible patients who are involved and interested in their sleep apnoea therapy.
References:
* ASV therapy is contraindicated in patients with chronic, symptomatic heart failure (NYHA 2-4) with reduced left ventricular ejection fraction (LVEF ≤ 45%) and moderate to severe predominant central sleep apnoea.
- Javaheri S., Smith J., Chung E., The Prevalence and Natural History of Complex Sleep Apnea, Journal of Clinical Sleep Medicine, Vol.5, No. 3, 2009.
- Mogri M et al. Hypoxemia in patients on chronic opiate therapy with and without sleep apnea. Sleep Breath, 2009;13:49-57.
- Karin G. Johnson and Douglas C. Johnson. Frequency of Sleep Apnea in Stroke and TIA Patients: A Meta-analysis. J Clin Sleep Med, 2010;6(2):131-137.