ASV-hoidon hyödyt
Muihin PAP-hoitoihin verrattuna ASV-hoidosta on merkittävää hyötyä sentraalisten unenaikaisten hengityshäiriöiden* hoidossa. Se parantaa AHI-indeksiä, vähentää hengitykseen liittyviä tapahtumia ja lievittää päiväaikaista väsymystä.
Kenelle ASV-hoito* soveltuu?
ASV-hoito soveltuu potilaille, joiden vasemman kammion ejektiofraktio (LVEF) on > 45 %.1,2,3,4
Voimme vahvistaa, että SERVE-HF-tutkimuksessa havaittu kuolleisuusriski koskee potilaita, joiden LVEF on ≤ 45 %, ja että ASV-hoidon haitat ovat yhteydessä olemassa olevaan vasemman kammion systoliseen vajaatoimintaan.5
Vasemman kammion ejektiofraktion pieneneminen on suljettava pois ennen ASV-hoidon aloittamista.1 Ennen ASV-hoitoa on tärkeää varmistaa, että LVEF on > 45 %. Tähän tarkoitukseen suositellaan sydämen kaikukuvausta.
Asiantuntijalausuntojen1,2,3,4 ja terveysviranomaisten mukaan ASV-hoitoa voidaan edelleen käyttää potilailla, joiden LVEF on > 45 % edellyttäen, että hoito on kliinisesti perusteltua. ASV-hoito soveltuu käytettäväksi seuraavissa tapauksissa: 1,2,3,4
- sydämen vajaatoiminta, jossa ejektiofraktio on säilynyt
- sentraalinen uniapnea, johon liittyy pitkäaikainen opioidihoito ilman keuhkoalveolien hypoventilaatiota
- idiopaattinen sentraalinen uniapnea tai Cheyne-Stokesin hengitys
- monitekijäinen (kompleksi)/ ilmaantuva / hoitoon reagoimaton sentraalinen uniapnea
- sentraalinen uniapnea iskeemisen aivohalvauksen jälkeen.
Toukokuusta 2015 lähtien Ranskan ja Saksan terveysviranomaiset ovat rajanneet vasta-aiheen sydämen vajaatoimintaan, johon liittyy pienentynyt ejektiofraktio (EF ≤ 45 %).4
Lue lisää ResMedin laitteesta, jolla annetaan ASV-hoitoa: AirCurve 10 CS PaceWave
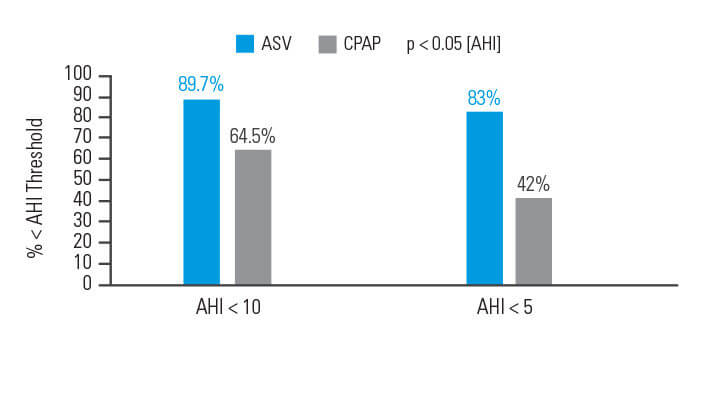
ASV-hoito tehoaa CPAP-hoitoa paremmin hengitykseen liittyviin tapahtumiin potilailla, jotka sairastavat kompleksia uniapneaa
Hoitoaikeen mukaisessa analyysissä (ITT) hoitovaste (apnea-hypopneaindeksi < 10) saavutettiin 90 päivän hoidon jälkeen 89,7 prosentilla ASV-hoitoa saaneista potilaista ja 64,5 prosentilla CPAP-hoitoa saaneista potilaista.6
[N = 66, prospektiivinen satunnaistettu tutkimus]
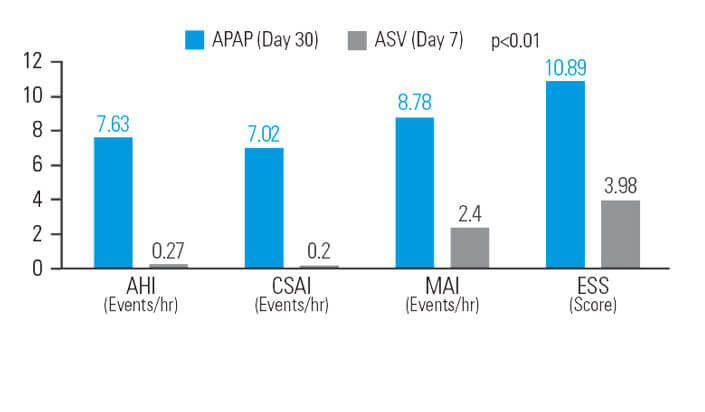
ASV-hoito vähentää APAP-hoidon jälkeen jäänyttä väsymystä potilailla, joilla on sekamuotoinen uniapnea
30 päivän APAP-hoidon jälkeen ASV-hoito pienensi apnea-hypopneaindeksiä (AHI) vielä (lähtötasoon verrattuna) 12,9 prosenttia lisää, sentraalisen apnean indeksiä (CSAI) 48,5 prosenttia lisää, mikroheräämisindeksiä (MAI) 26,1 prosenttia lisää ja Epworthin uneliaisuusasteikkoa (ESS) 37,9 prosenttia lisää, kun käytössä oli vastaava keskipaine.7
[N = 42, sekventiaalinen tutkimus]
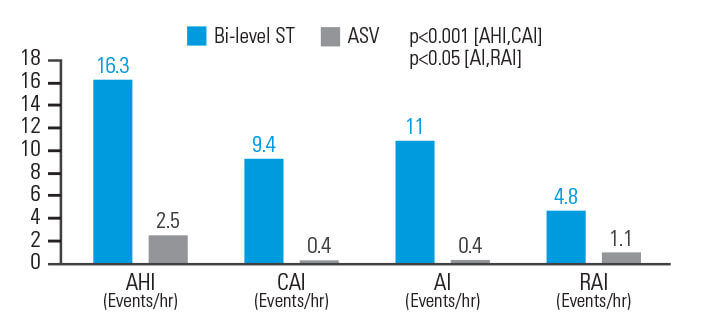
ASV-hoito vähensi hengitykseen liittyviä tapahtumia tehokkaammin kuin ST-kaksoispainehoito potilailla, joilla oli opioidien aiheuttama sentraalinen uniapnea
Potilailla, joilla oli opioidien aiheuttama sentraalinen uniapnea, ASV-hoito pienensi AHI-indeksiä 84,7 %, sentraalisen apnean indeksiä (CAI) 95,7 %, apneaindeksiä (AI) 96,4 % ja hengityshäiriöistä johtuvien heräämisten indeksiä (RAI) 77,1 % ST-kaksoispainehoitoon verrattuna. Hengitysparametrit normalisoituivat 83,3 prosentilla ASVAuto-hoitoa saaneista potilaista, mutta vain 33,3 prosentilla ST-kaksoispainehoitoa saaneista potilaista.8
[N = 18, prospektiivinen, satunnaistettu, vaihtovuoroinen unipolygrafiatutkimus]
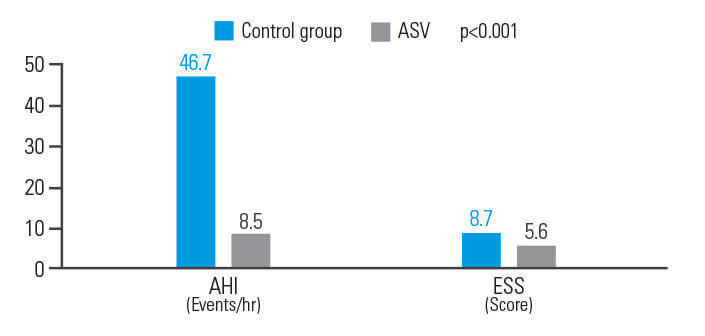
ASV parantaa AHI- ja ESS-arvoja potilailla, joilla on ollut akuutti iskeeminen aivohalvaus
ASV-hoito paransi hoitotuloksia sentraalista uniapneaa sairastavilla potilailla, joilla on ollut akuutti iskeeminen aivohalvaus. Se pienensi AHI-indeksiä 81,8 prosenttia ja ESS-arvoa 35,6 prosenttia.9
[N = 15, yhden keskuksen retrospektiivinen analyysi]
ASV parantaa sydämen minuuttitilavuutta ja ennustetta potilailla, joilla on sydämen vajaatoiminta, jossa ejektiofraktio on säilynyt*
Prospektiiviset satunnaistetut ja havainnoivat sydämen vajaatoimintaa koskevat esitellyt ja pian julkaistavat tutkimukset viittaavat siihen, että ASV-hoidosta voi olla hyötyä potilaille, joilla on sydämen vajaatoiminta, jossa ejektiofraktio on säilynyt, ja potilaille, joilla on sentraalinen uniapnea ja samanaikainen obstruktiivinen uniapnea.10 Tällä hetkellä ei ole mitään näyttöä siitä, että ASV-hoito aiheuttaisi näille potilaille mitään riskiä.
ASV-videot: katso asiantuntijakeskustelua ASV-hoidosta.
Lisätietoja hoitovaihtoehdoista

Sentraalisen uniapnean hoitovaihtoehdot
Adaptiivinen servoventilaatio (ASV)* on sopiva valinta useimpien monimutkaisempien potilaiden hoitamiseen.

OSA:n hoitovaihtoehdot
ResMedillä on laaja valikoima obstruktiivisen uniapnean hoitotuotteita kuten CPAP-, APAP ja kaksoispainelaitteet ja hammaskiskot.

Hoidon seuranta
ResMed Air Solutions -sarjaan kuuluva AirView-sovellus tarjoaa mahdollisuuden edistyksellisiin hoitoa koskeviin näkemyksiin ja aivan uudenlaisen mahdollisuuden hoitoratkaisuihin myös etänä.
Viitteet:
* ASV-hoito on vasta-aiheista potilailla, joilla on krooninen oireinen sydämen vajaatoiminta (NYHA 2–4), pienentynyt vasemman kammion ejektiofraktio (LVEF ≤ 45 %) ja keskivaikea tai vaikea pääasiallisesti sentraalinen uniapnea.
- d’Ortho et al. European Respiratory & Pulmonary Diseases, 2016;2(1):Epub ahead of print. http://doi.org/10.17925/ERPD.2016.02.01.1.
- Priou P & al. Adaptive servo-ventilation: How does it fit into the treatment of central sleep apnoea syndrome? Expert opinions. Revue des Maladies Respiratoires, 2015 Dec, 32(10):1072-81.
- Aurora RN & al. Updated Adaptive Servo-Ventilation Recommendations for the 2012 AASM Guideline: ”The Treatment of Central Sleep Apnea Syndromes in Adults: Practice Parameters with an Evidence-Based Literature Review and Meta-Analyses”. Journal of Clinical Sleep Medicine, 2016 May 15, 12(5):757-61.
- Randerath W et al. ERJ Express. Published on December 5, 2016 as doi: 10.1183/13993003.00959-2016.
- AirCurve 10 CS PaceWave clinical manual July 2015.
- Morgenthaler et al. The Complex Sleep Apnea Resolution Study, Sleep, Vol. 37, No. 5, 2014.
- Su et al. Adaptive pressure support servoventilation: a novel treatment for residual sleepiness associated with central sleep apnea events, Sleep Breath, 2011;15:695-699.
- Cao et al. A Novel Adaptive Servoventilation (ASVAuto) for the Treatment of Central Sleep Apnea Associated with Chronic Use of Opioids, Journal of Clinical Sleep Medicine, Vol. 10, No. 8, 2014.
- Brill et al. Adaptive servo-ventilation as treatment of persistent central sleep apnea in post-acute ischemic stroke patients, Sleep Medicine 15, 2014;1309-1313.
- Bitter T et al. Eur Respir J 2010; 36: 385–392 06 Yoshihisa et al. European Journal of Heart Failure doi:10.1093/eurjhf/hfs197